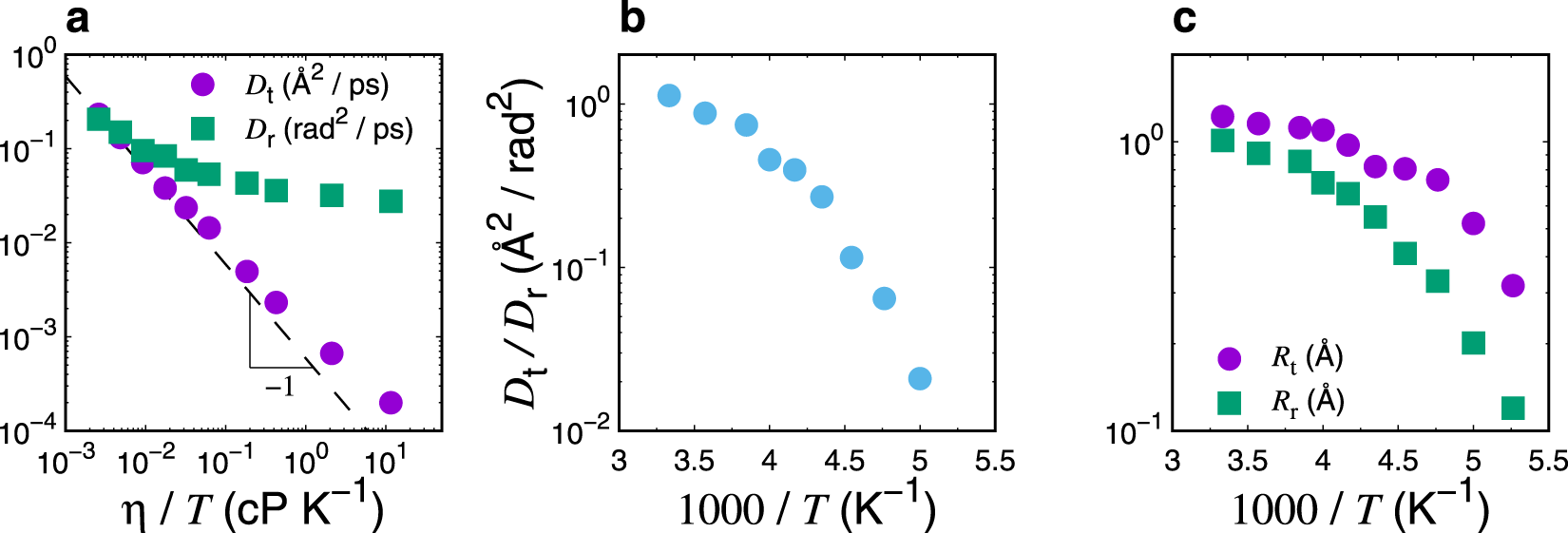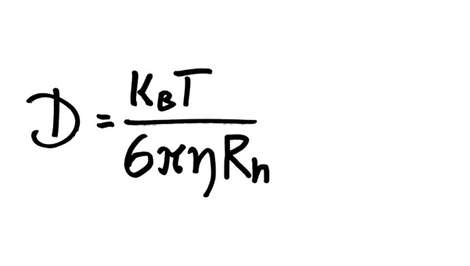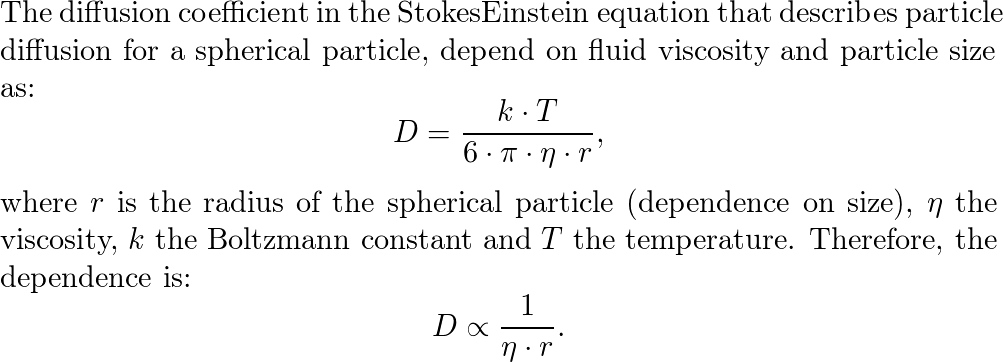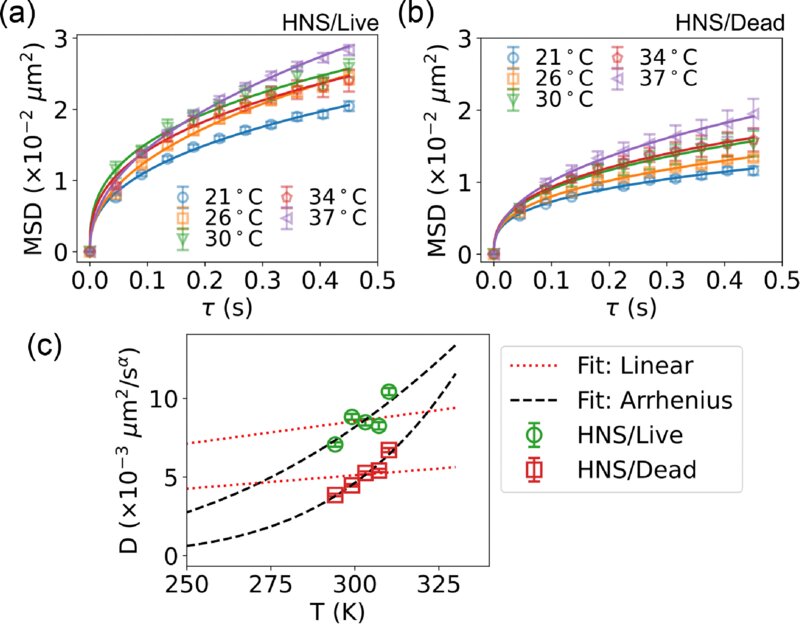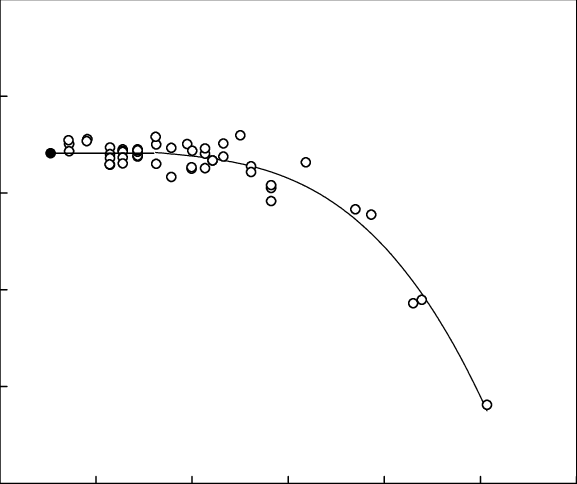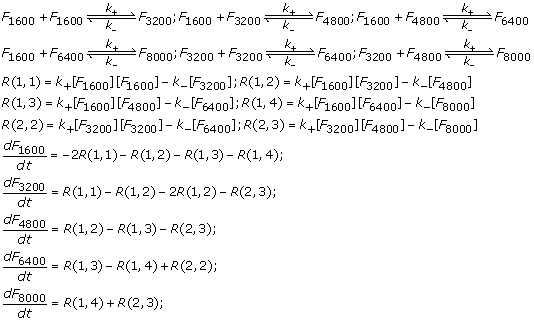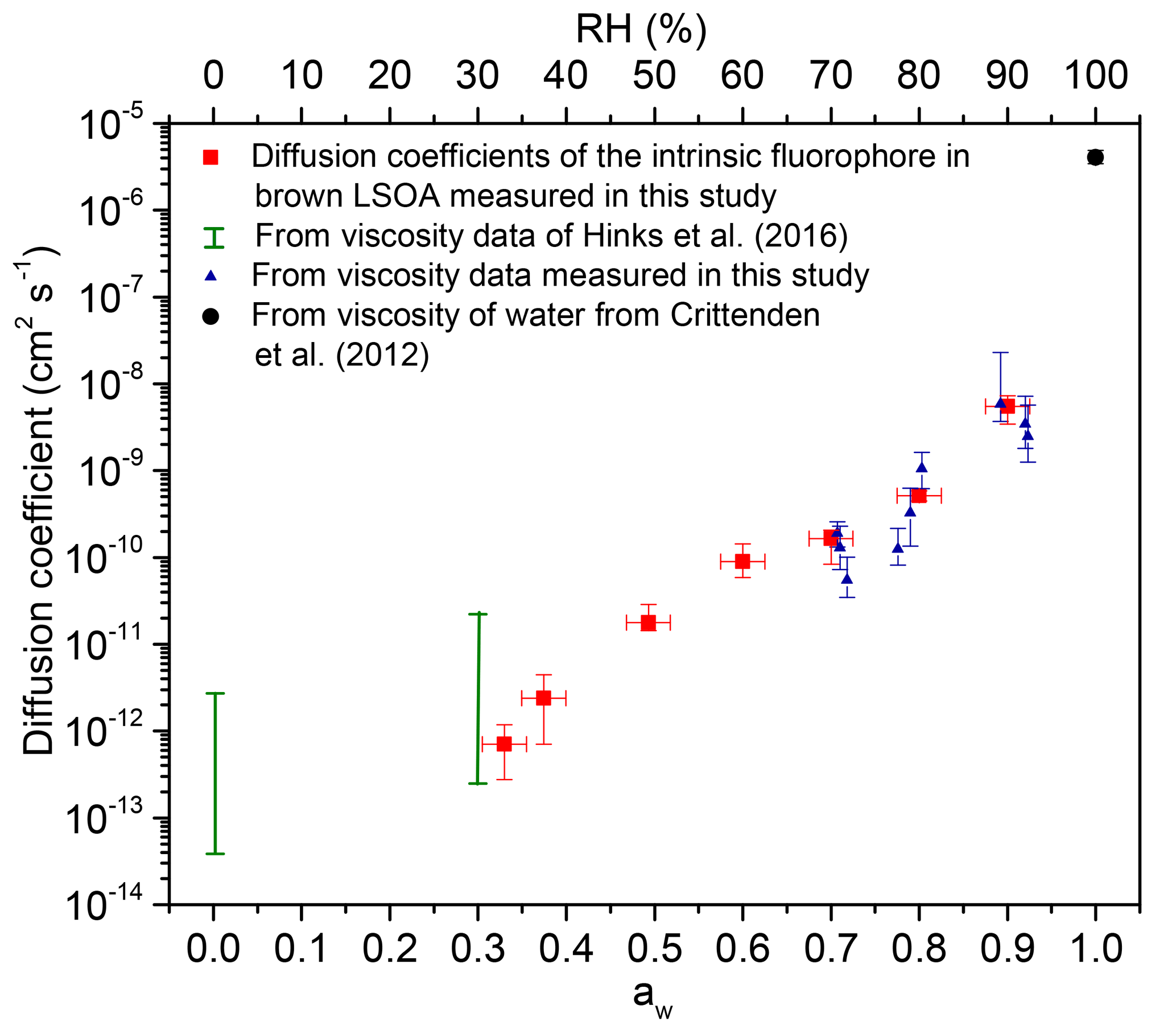
ACP - Viscosities, diffusion coefficients, and mixing times of intrinsic fluorescent organic molecules in brown limonene secondary organic aerosol and tests of the Stokes–Einstein equation

SOLVED: The Stokes-Einstein relationship defines the diffusion constant as the ratio of thermal (kinetic) movement of a spherical particle to the resistance to movement due to solvent viscosity: kT D = 6xnR

The Stokes-Einstein equation and the diffusion of ferrocene in imidazolium-based ionic liquids studied by cyclic voltammetry: Effects of cation ion symmetry and alkyl chain length - ScienceDirect
![PDF] Viscosities, diffusion coefficients, and mixing times of intrinsic fluorescent organic molecules in brown limonene secondary organic aerosol and tests of the Stokes–Einstein equation | Semantic Scholar PDF] Viscosities, diffusion coefficients, and mixing times of intrinsic fluorescent organic molecules in brown limonene secondary organic aerosol and tests of the Stokes–Einstein equation | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/222d3d4697fedfb1d5c1adba7df3adf65f14adad/17-Figure5-1.png)
PDF] Viscosities, diffusion coefficients, and mixing times of intrinsic fluorescent organic molecules in brown limonene secondary organic aerosol and tests of the Stokes–Einstein equation | Semantic Scholar

Fick's Law of Diffusion.pdf - Stokes-Einstein Equation D = Diffusion coefficient K = Boltzmanns constant T = absolute temperature K r = molecular | Course Hero

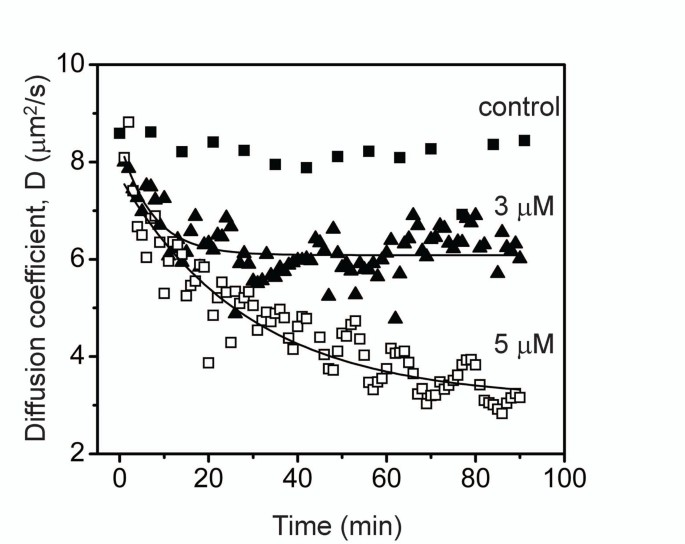
SOLVED: Using the Stokes-Einstein equation and the Wilkie-Chang correlation (eq: (6.6.25), see below), estimate the diffusion coefficient of oxygen in water at 298K. Take the molecular diameter to have a value of

The Stokes-Einstein equation gives an estimate of the frictional coefficient, f, in terms of molecular

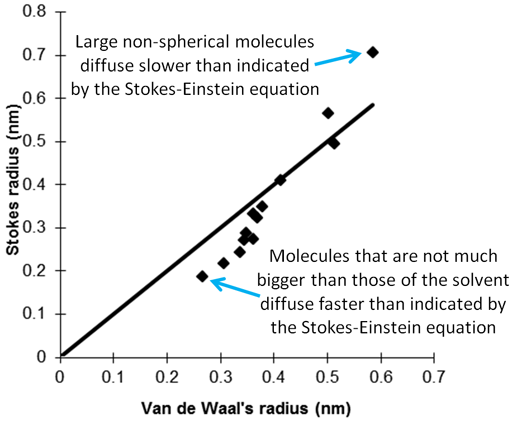
Diffusion Coefficient (D) and Stokes−Einstein Hydrodynamic Radius (r H... | Download Scientific Diagram