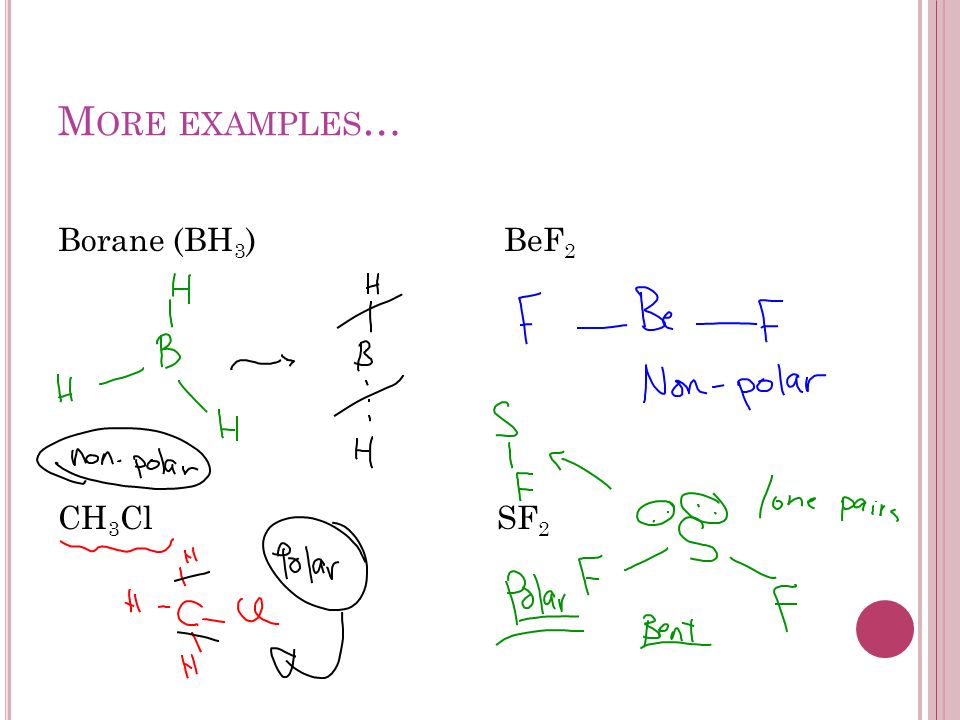
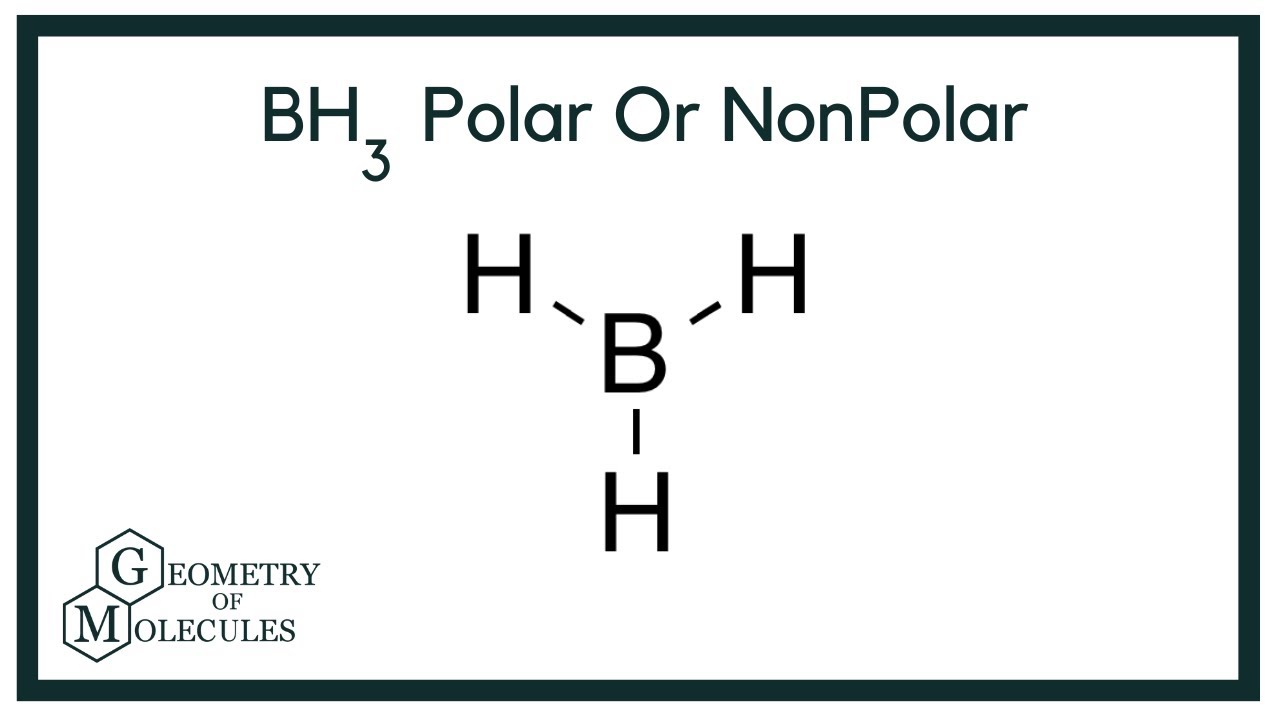
BH3 lewis structure, molecular geometry, polar or nonpolar, hybridization, Bond angle | Molecular geometry, Covalent bonding, Molecular

SOLVED: Question 8 Determine if this molecule (BH3) is polar or nonpolar. Hot yet answered. Points out of 1.00. Flag question. H-B-H Select one: a. It cannot be determined. b. Nonpolar because
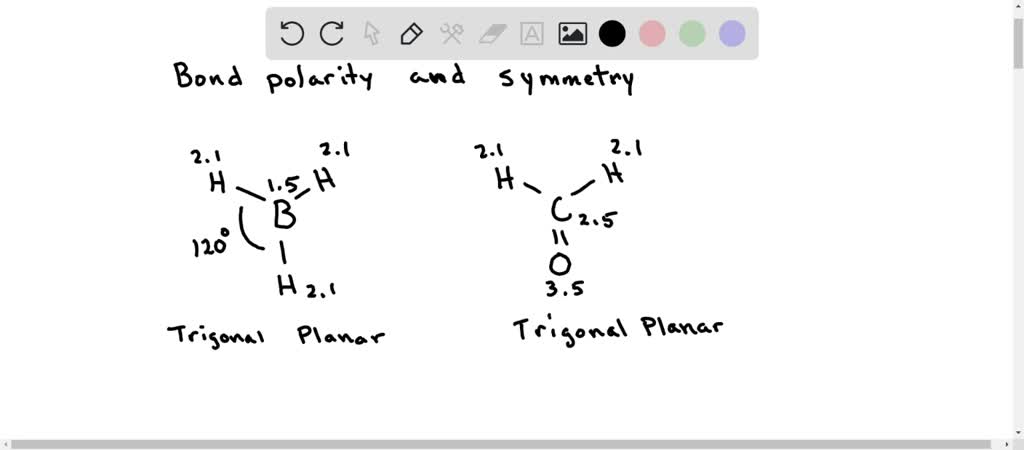
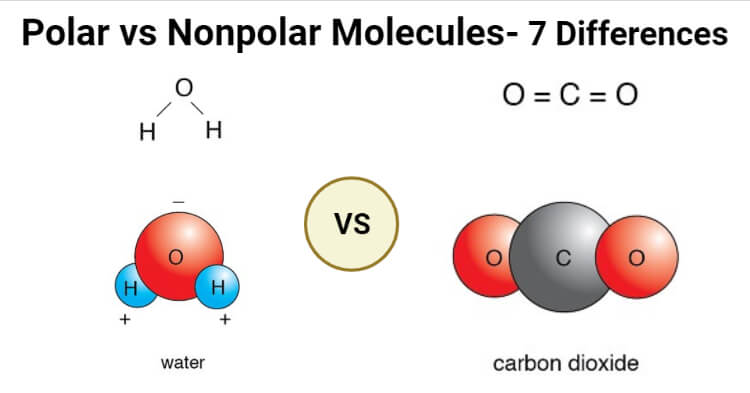
SOLVED: Although BH3 and CH2O have similar shapes, one is polar and the other is non-polar. Explain why.
![SOLVED: Classify each molecule as polar or nonpolar: CHCl3 [Select ] CH2OCH2 [Select ] [Select ] CH3CH2CH3 [Select ] B2H6 SOLVED: Classify each molecule as polar or nonpolar: CHCl3 [Select ] CH2OCH2 [Select ] [Select ] CH3CH2CH3 [Select ] B2H6](https://cdn.numerade.com/ask_images/280774a7adb34fc9baeeb5c3a82ac8f2.jpg)
SOLVED: Classify each molecule as polar or nonpolar: CHCl3 [Select ] CH2OCH2 [Select ] [Select ] CH3CH2CH3 [Select ] B2H6