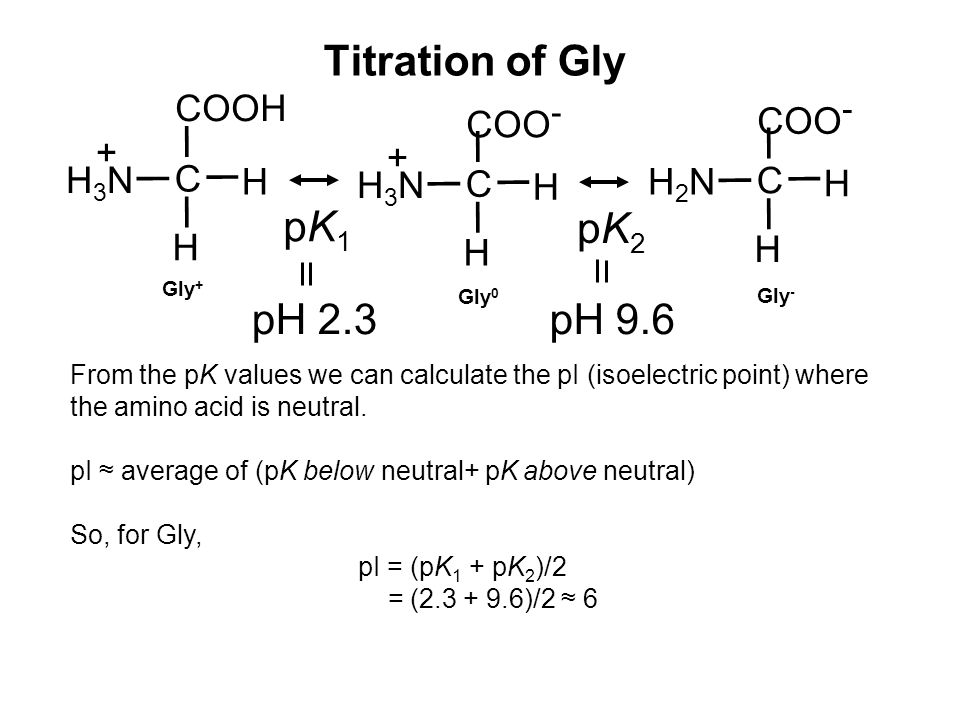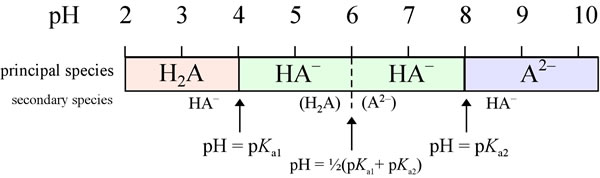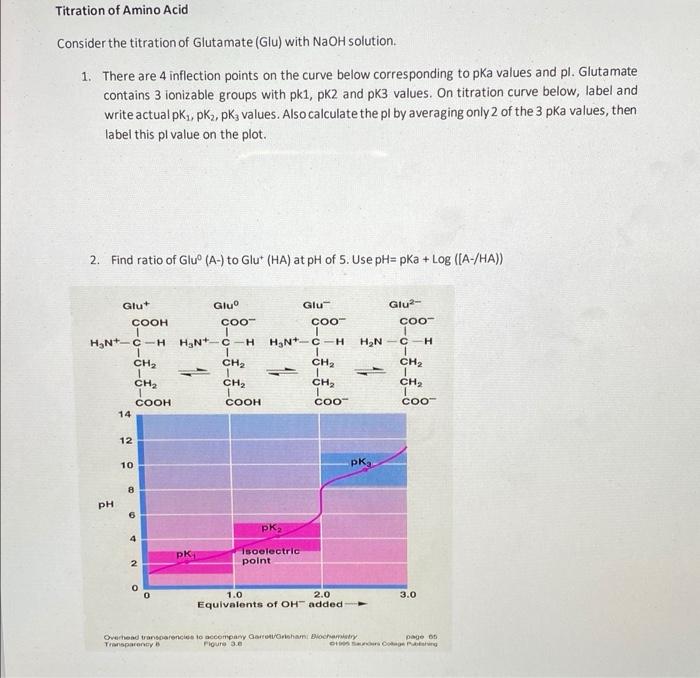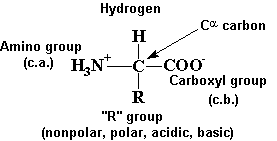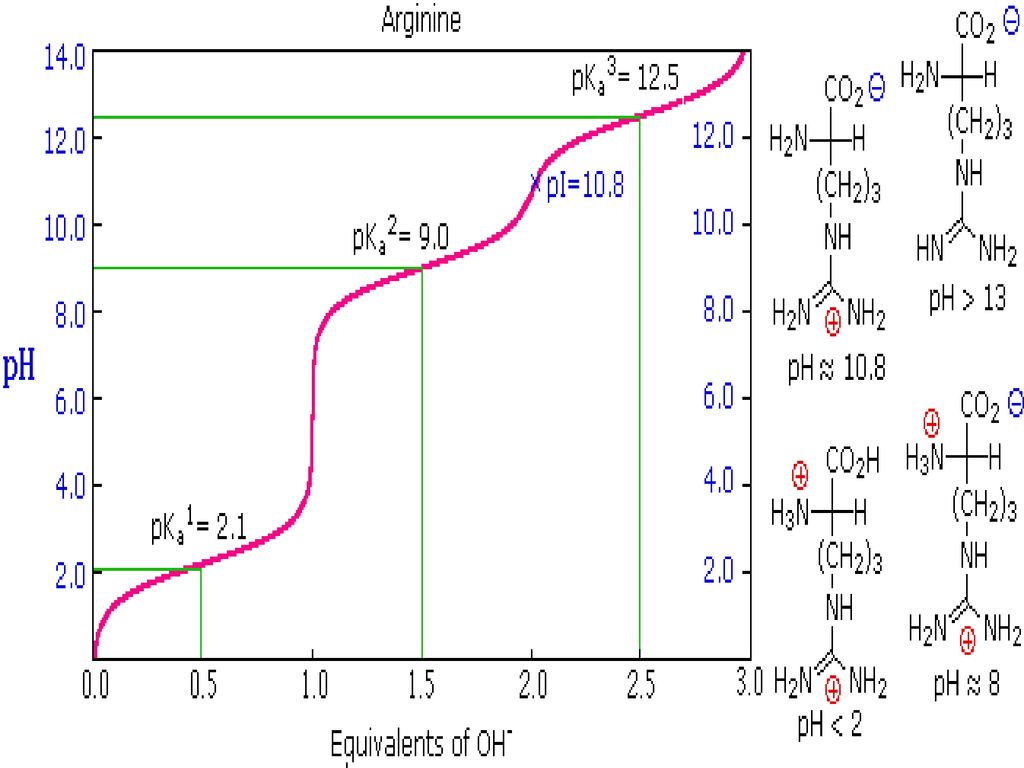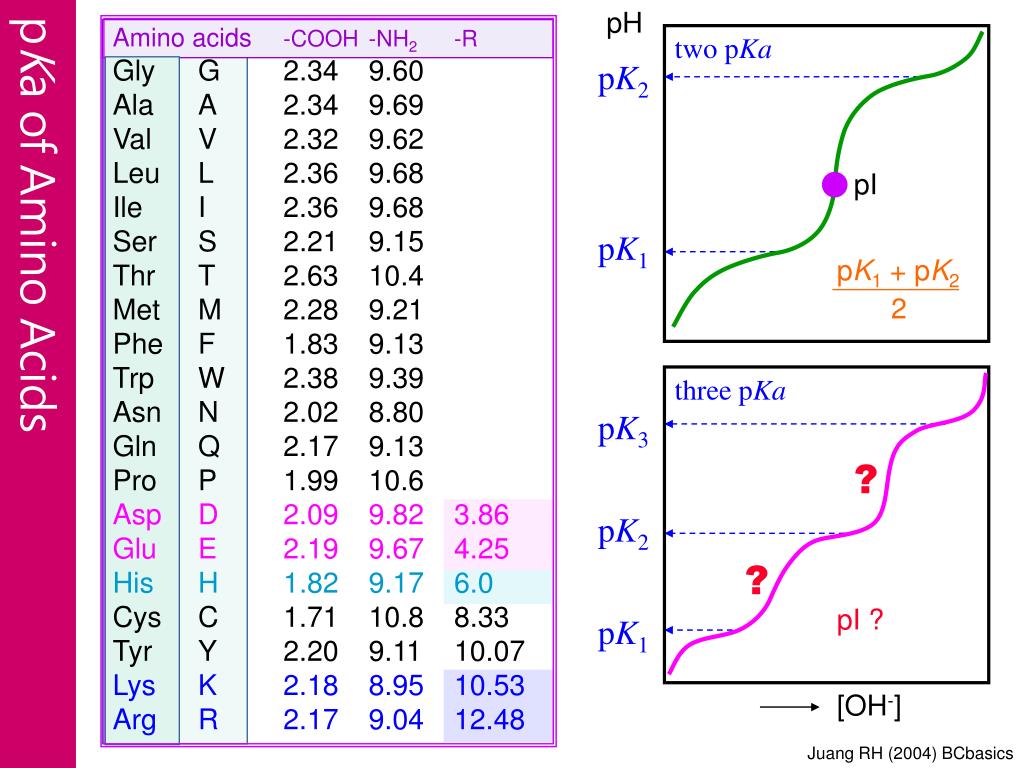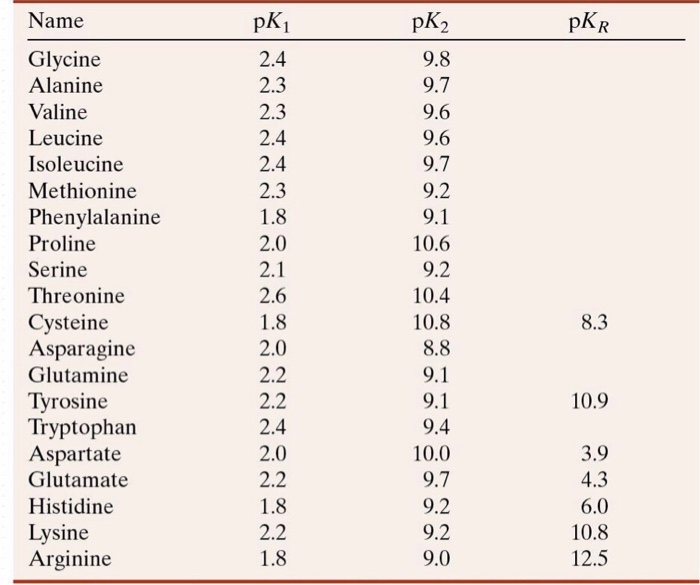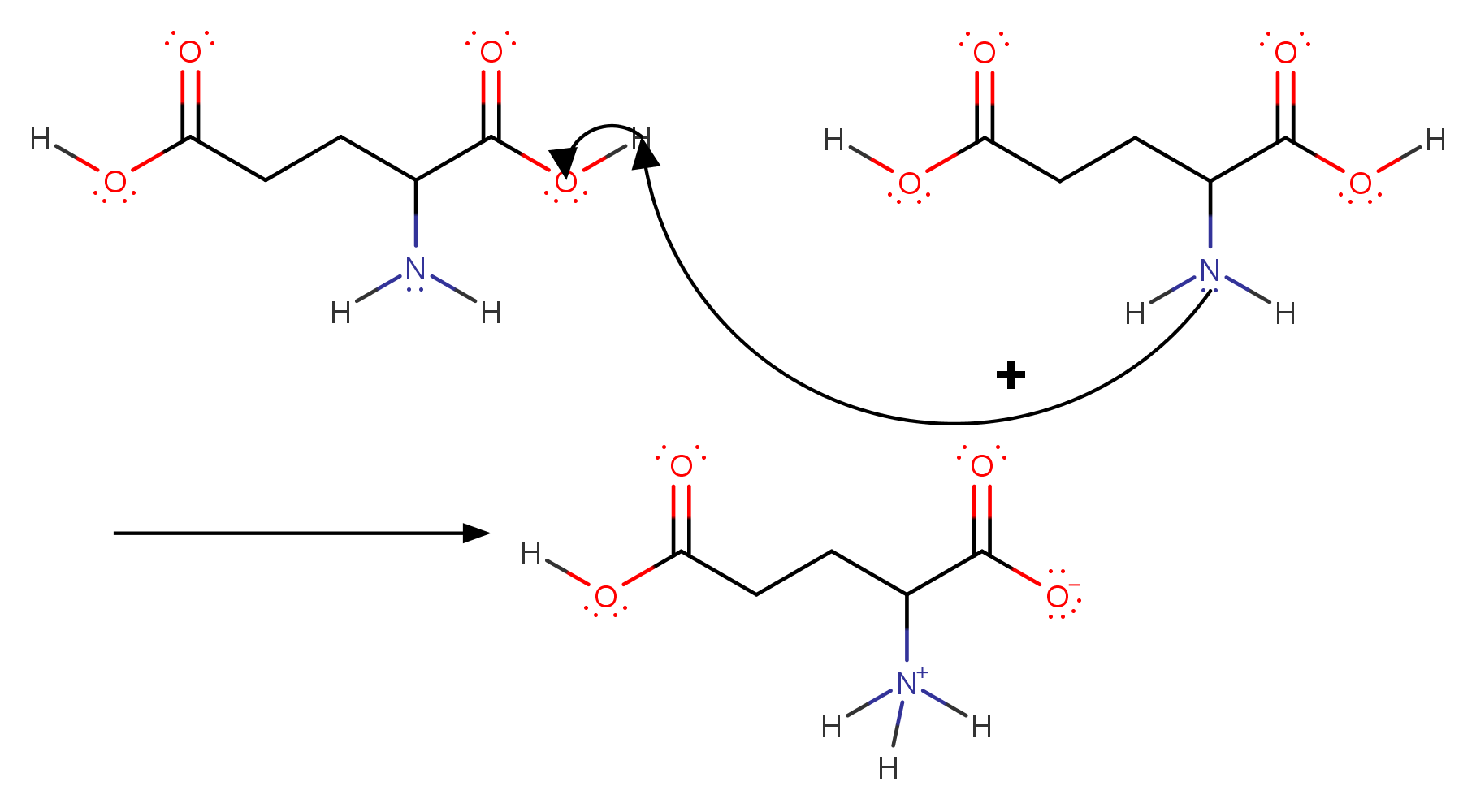
Using the Pka' Values for glutamic acid (pk1= 2.19, pk2= 9.67, pkR= 4.25) indicate the ionic form which predominates at: a) pH 1.0 b)pH 7.0 c) pH13 d) What is the net
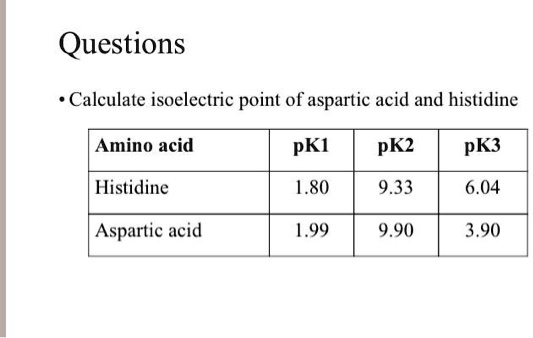
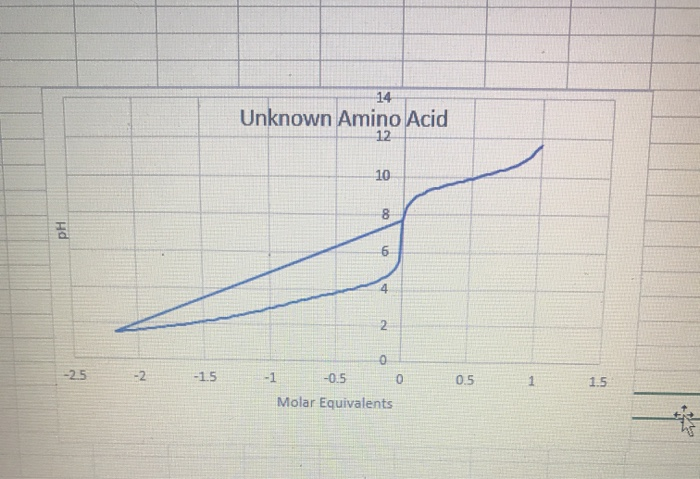
Calculate the isoelectric point of aspartic acid and histidine. Amino acid pKI pK2 pK3 Histidine 1.80 9.33 6.04 Aspartic acid 1.99 9.90 3.90