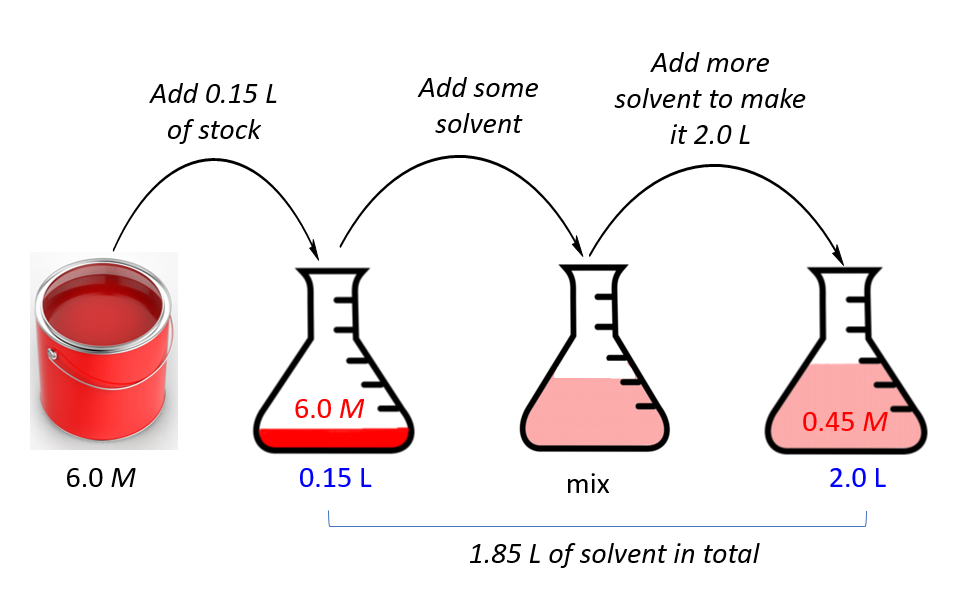
SOLVED: make a stock solution calculation for an ending volume of 100 mL / prepare the stock in a 250 mL volumetric flask Which of the following equations demonstrates the correct initial

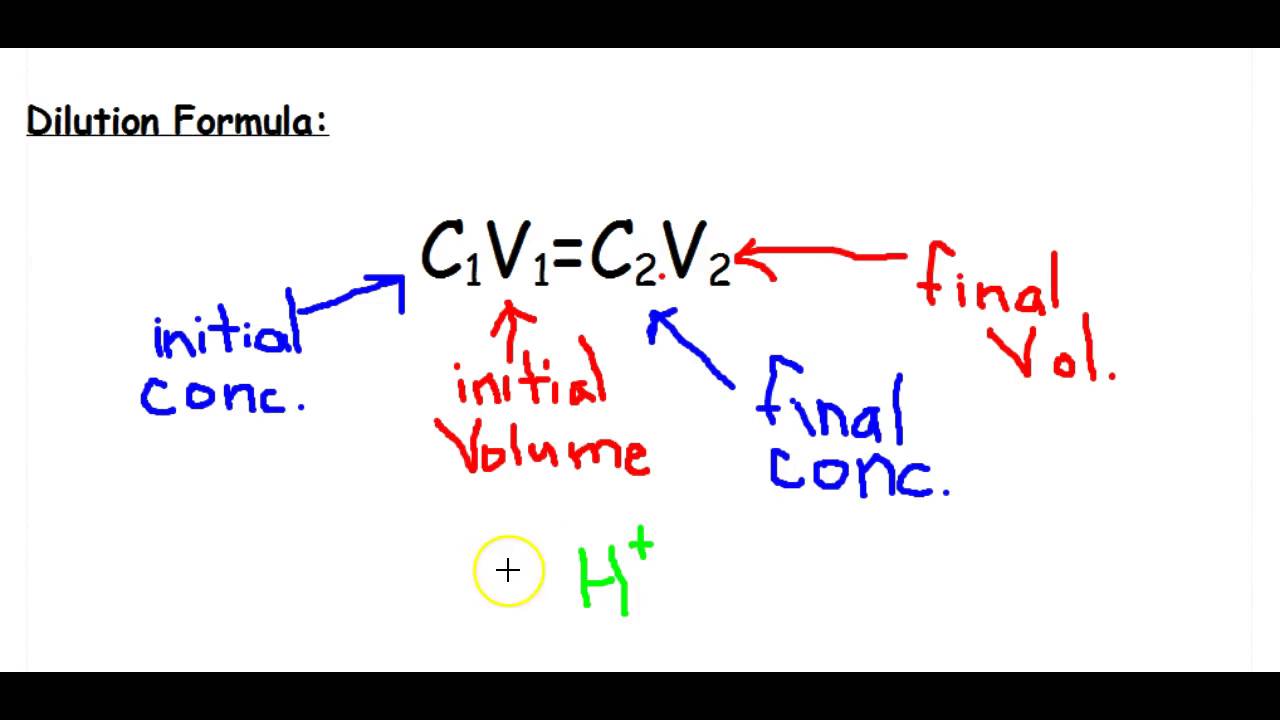
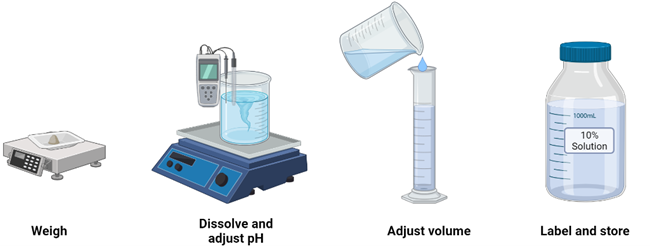
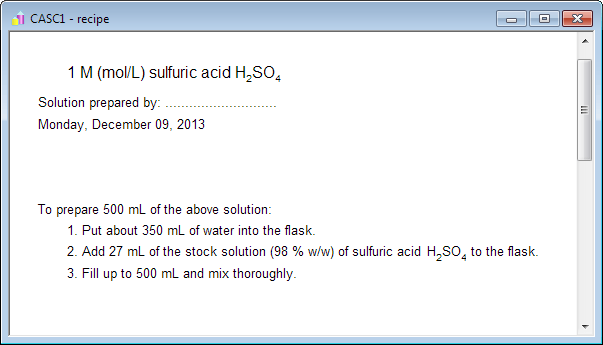
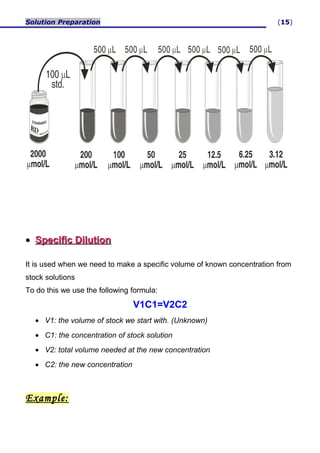
Stock Solution| Dilution Formula| How to prepare Stock Solutions|Numeric... | Solutions, Preparation, Formula

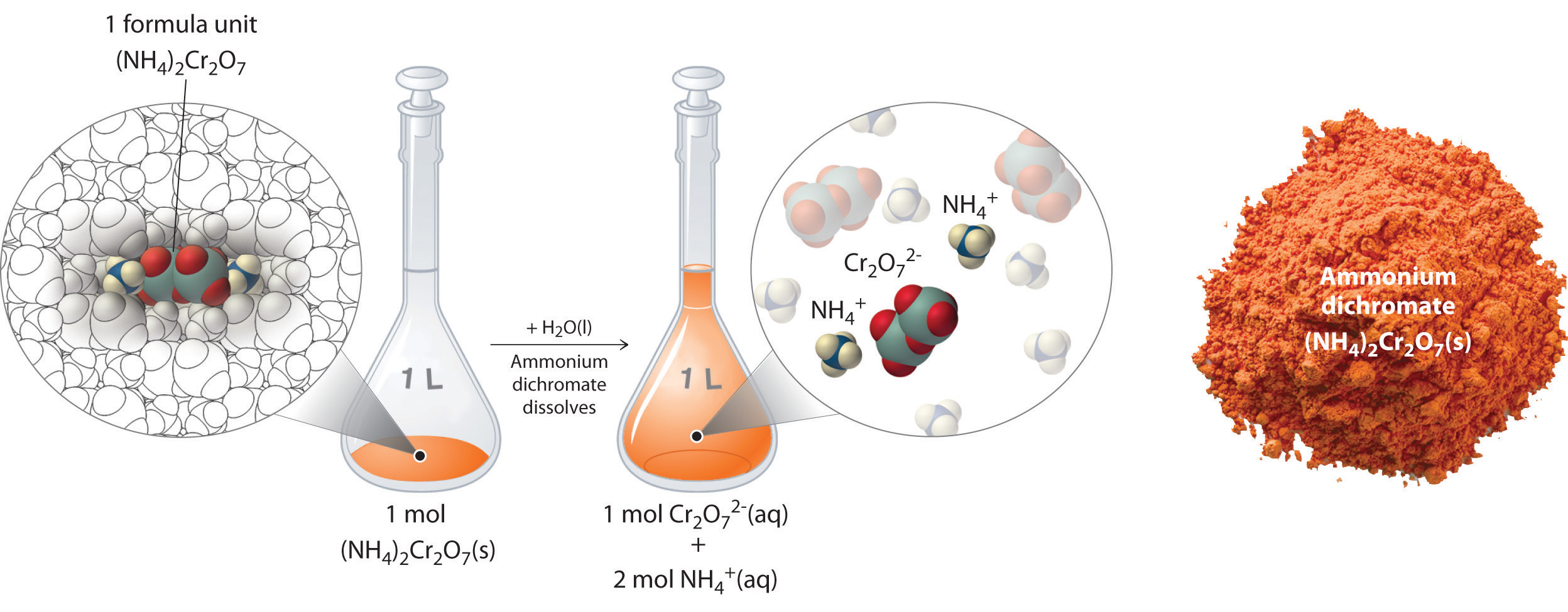
Solutions Molarity = concentration term used to describe an amount of solute dissolved in a given amount of solvent. Concentrated solution = contains large.
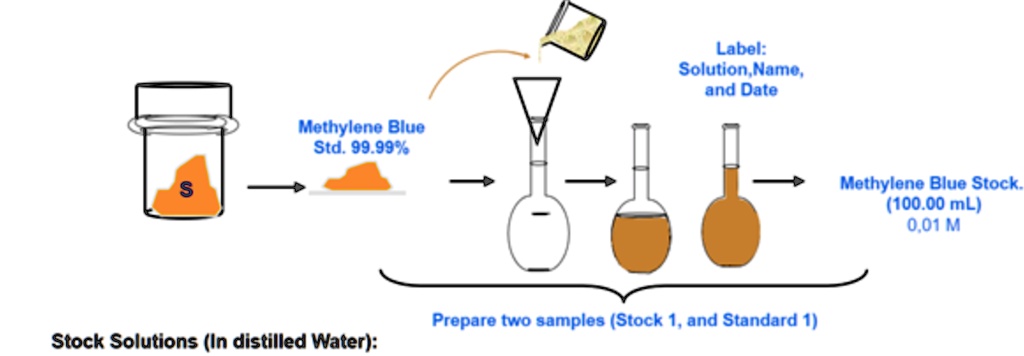
SOLVED: 'Calculate the weight of methylene blue to prepare the stock solution in the case of the illustration. WM: 319,85 g/mol Methylene blue Label: Solution Name, and Date Methylene Blue Std. 99.99%8
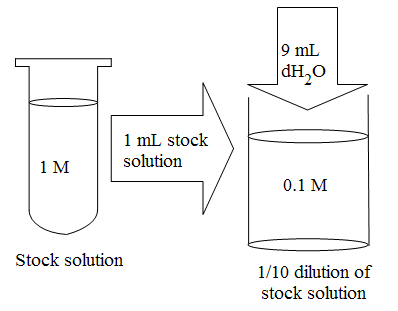


![ANSWERED] Calculate the volume of a stock solution of Ba(OH)2, with a - Kunduz ANSWERED] Calculate the volume of a stock solution of Ba(OH)2, with a - Kunduz](https://media.kunduz.com/media/sug-question/raw/56683410-1659453625.5455317.jpeg)